“`html
Boron Nitride Ceramic Substrate for Chemical Applications: A Comprehensive Technical Guide
In the demanding landscape of modern chemical processing, material selection is the bridge between experimental success and industrial failure. Among the elite class of technical ceramics, Boron Nitride (BN) has emerged as a critical material for high-performance substrates. Often referred to as “White Graphite” due to its lubricity and hexagonal crystal structure, Boron Nitride ceramic substrates offer a unique combination of thermal, electrical. And chemical properties that are unattainable with traditional materials like alumina or quartz.
Great Ceramic specializes in the precision engineering of Boron Nitride ceramic substrates for chemical environments, providing custom-machined solutions that withstand the most corrosive and thermally volatile conditions. This guide explores the technical intricacies of BN substrates, their behavior in chemical systems. And the advanced CNC machining processes required to bring these components to life.
The Science of Boron Nitride Ceramic Substrates
Boron Nitride is a synthetic ceramic material, typically produced in two primary forms: hexagonal (h-BN) and cubic (c-BN). For chemical substrates, hexagonal Boron Nitride is the industry standard. It is produced through hot-pressing or pyrolytic deposition, resulting in a material that is highly anisotropic and chemically inert.
Material Grades and Varieties
When selecting a Boron Nitride ceramic substrate for chemical use, it is essential to understand the distinction between grades:
- Hot-Pressed Boron Nitride (HBN): This is the most common form, produced by pressing BN powder at high temperatures. It often contains a small amount of boric oxide (B2O3) as a binder. This can influence chemical resistance.
- Pyrolytic Boron Nitride (PBN): Produced via Chemical Vapor Deposition (CVD), PBN is 99.99% pure. It has no binder, making it the ultimate choice for high-purity chemical applications where outgassing or contamination must be zero.
- Composite BN: Often mixed with Silicon Carbide (SiC) or Nitrure d'aluminium (AlN) to enhance hardness or thermal expansion matching.
Tableaux des propriétés techniques
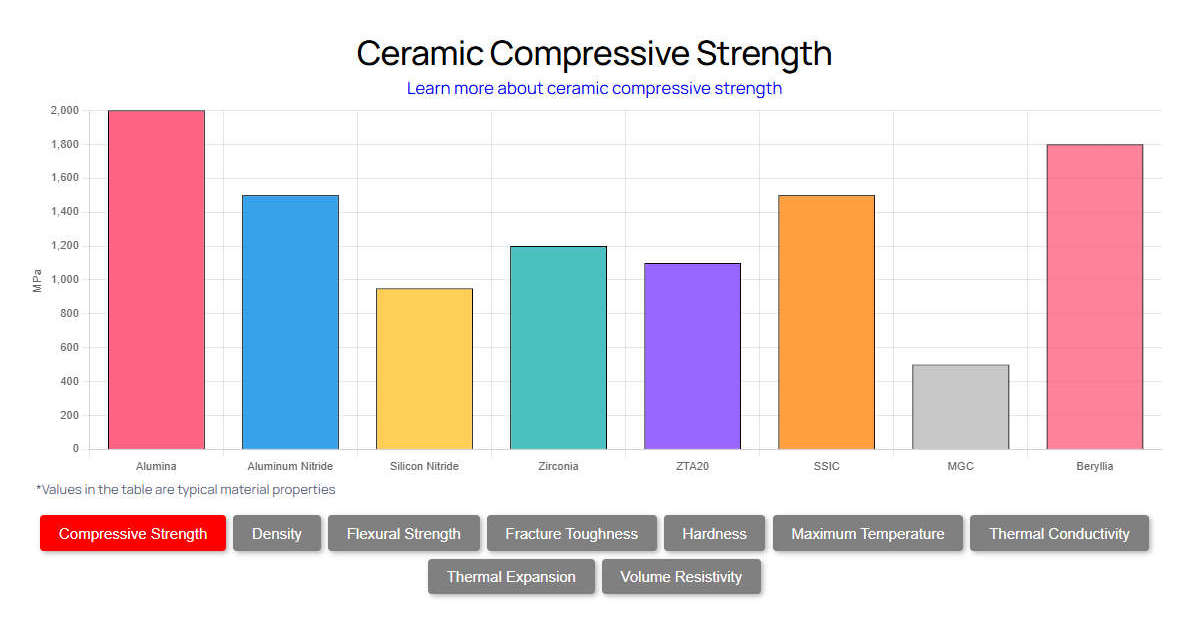
To design effective chemical systems, engineers require precise data. The following tables outline the physical and thermal characteristics of standard hot-pressed Boron Nitride ceramic substrates.
Tableau 1 : Propriétés physiques et mécaniques
| Propriété | Unité | Value (Typical HBN) | Value (PBN) |
|---|---|---|---|
| Densité | g/cm³ | 1.9 – 2.1 | 2.15 – 2.20 |
| Résistance à la flexion | MPa | 50 – 100 | 150 – 200 (Parallel) |
| Résistance à la compression | MPa | 100 – 150 | 250+ |
| Module d'élasticité | GPa | 50 – 80 | 100 – 200 |
| Dureté (Mohs) | - | 2.0 | 2.0 |
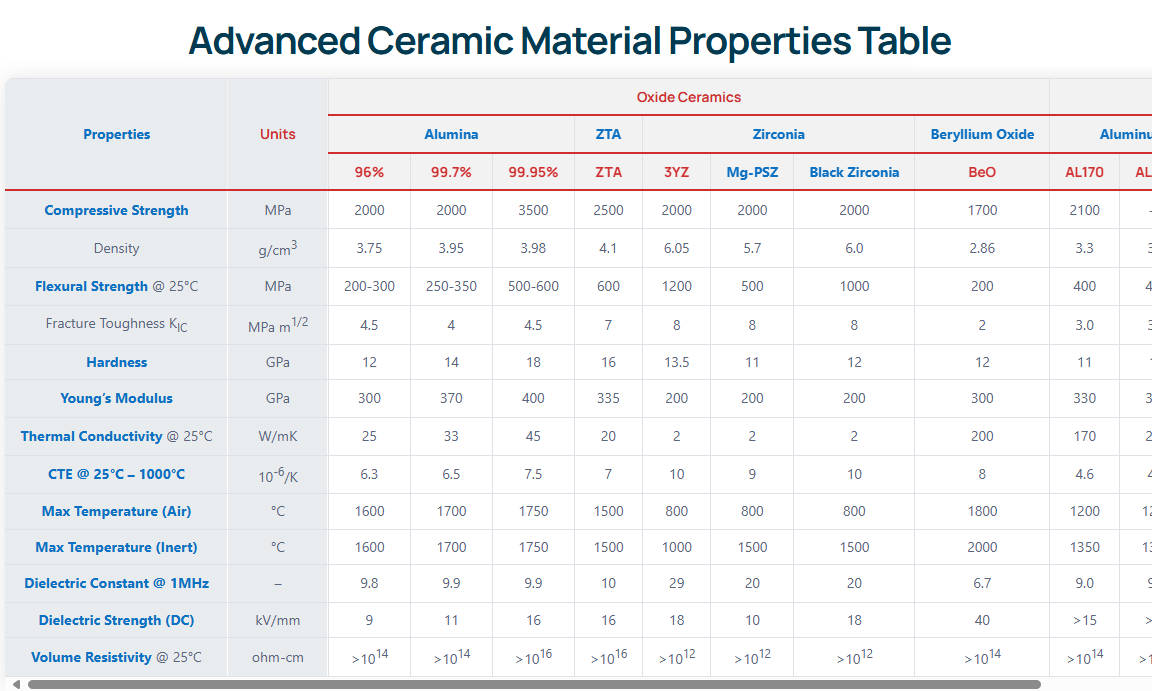
Table 2: Thermal and Electrical Properties
| Propriété | Unité | Value (Typical HBN) | Value (PBN) |
|---|---|---|---|
| Thermal Conductivity (25°C) | W/m-K | 30 - 60 | 60 (Perp) / 200+ (Parallel) |
| Thermal Expansion Coeff. | 10-⁶/°C | 0.5 – 4.0 | 0.5 – 3.0 |
| Max Operating Temp (Inert) | °C | 2000 | 2200 |
| Max Operating Temp (Air) | °C | 900 | 1000 |
| Rigidité diélectrique | kV/mm | 20 – 40 | 50 – 100 |
Chemical Resistance and Stability
The primary reason for utilizing a nitrure de bore ceramic substrate for chemical processes is its extraordinary chemical stability. BN is non-wetting to most molten metals and glasses. And it remains stable in the presence of many aggressive reagents.
Resistance to Molten Metals
Unlike alumina or zircone, Boron Nitride is not “wetted” by molten aluminum, magnesium, zinc, or lead. This makes it an ideal substrate for chemical sensors or electrodes submerged in molten metal baths. It prevents the formation of intermetallic layers that typically degrade other ceramic substrates.
Acid and Base Interaction
BN is generally resistant to most organic solvents and many inorganic acids. However, its behavior depends on the grade. Hot-pressed BN with a boric oxide binder may show slight degradation in the presence of strong boiling acids or alkaline solutions as the binder leaches out. For these specific chemical environments, Great Ceramic recommends PBN or high-purity HBN with stabilized binders.
Oxidation and Atmosphere
While BN can withstand temperatures exceeding 2000°C in vacuum or nitrogen atmospheres, it begins to oxidize in air at approximately 850°C – 1000°C. During oxidation, a thin layer of B2O3 forms on the surface. This can provide a temporary protective barrier but may change the chemical purity of the environment.
Industry Applications for BN Ceramic Substrates
The convergence of electrical insulation and thermal conductivity makes BN substrates indispensable across several high-tech chemical sectors.
1. Semiconductor and Wafer Processing
In the production of semiconductors, Boron Nitride substrates are used in Ion Implantation and MOCVD (Metal-Organic Chemical Vapor Deposition) systems. The high purity of PBN prevents wafer contamination, while the thermal conductivity ensures uniform temperature distribution during chemical vapor reactions.
2. Chemical Sensors and Electrochemistry
BN substrates serve as the base for thin-film electrodes in high-temperature chemical sensors. Their ability to maintain high electrical resistivity even at 1000°C allows for accurate signal measurement in corrosive gas environments where standard FR4 or alumina substrates would fail or become conductive.
3. Molten Metal Handling and Crucibles
In analytical chemistry involving molten metals, BN substrates act as support plates or thermal breaks. Because BN does not react with the melt, it ensures the chemical integrity of the alloy or sample being analyzed.
4. High-Vacuum Chemical Systems
Due to its low vapor pressure, Boron Nitride does not outgas significantly in high-vacuum environments. This is vital for chemical research involving mass spectrometry or molecular beam epitaxy (MBE), where even a few parts-per-million of stray gas can ruin an experiment.
CNC Machining of Boron Nitride Substrates
One of the most significant advantages of Boron Nitride, particularly for custom chemical applications, is its machinability. While most ceramics require diamond grinding and are prone to brittle fracture, BN is often described as “machinable ceramic.”
Capacités d'usinage de précision
At Great Ceramic, we leverage advanced CNC technology to produce complex geometries that would be impossible with other high-performance ceramics. Boron Nitride can be turned, milled, drilled. And threaded using standard high-speed steel or carbide tooling, though specialized techniques are required to maintain tolerances and surface integrity.
Technical Considerations for BN Machining:
- Tolerances: We consistently achieve tolerances of ±0.01mm. This is critical for substrates that must fit into precision chemical reactors or sensor housings.
- Finition de la surface : For chemical applications, a smooth surface finish is often required to prevent reagent trapping. We can achieve surface roughness (Ra) values as low as 0.4 μm.
- Brittleness: Despite its softness, BN is still a ceramic. Sharp corners should be avoided in designs. radiused edges help prevent chipping during the machining process and in final use.
- Anisotropy: Especially in hot-pressed BN, the properties vary depending on the direction of the press. Great Ceramic’s engineers consider the grain orientation of the BN block relative to the substrate’s thermal load to maximize performance.
Why Choose Machined BN over Molded Ceramics?
Molding or dry-pressing ceramic parts often leads to density variations and the need for expensive tooling. CNC machining from high-quality BN blanks allows for rapid prototyping and the production of low-to-medium volumes without the upfront cost of molds. This is particularly beneficial for custom chemical labware and specialized substrate designs.
Comparison: BN vs. Alumine vs. Aluminum Nitride
Choosing the right substrate requires a comparative look at the “big three” technical ceramics.
| Fonctionnalité | Nitrure de bore (BN) | Alumine (Al2O3) | Nitrure d'aluminium (AlN) |
|---|---|---|---|
| Conductivité thermique | High (30-200 W/mK) | Moderate (25-30 W/mK) | Very High (170-200 W/mK) |
| Usinabilité | Excellent (CNC) | Poor (Grinding only) | Poor (Grinding only) |
| Résistance aux chocs thermiques | Excellent | Modéré | Bon |
| Inertie chimique | Superior (Non-wetting) | Bon | Sensitive to moisture |
| Coût | Haut | Faible | Medium-High |
While Aluminum Nitride offers higher thermal conductivity in some cases, its susceptibility to hydrolysis (reacting with water/moisture) makes it less ideal for many chemical applications compared to the robust stability of Boron Nitride.
Foire aux questions (FAQ)
1. Is Boron Nitride ceramic substrate toxic for chemical use?
No, Boron Nitride is chemically inert and non-toxic. It is often used in cosmetic and pharmaceutical manufacturing equipment because it does not react with organic compounds or biological tissues.
2. Can Boron Nitride substrates be used in contact with strong acids?
BN is resistant to most cold acids. However, hot phosphoric acid or strong alkaline solutions can attack the material, especially if binders are present. For extreme acid resistance, Pyrolytic Boron Nitride (PBN) is the recommended grade.
3. How does moisture affect BN substrates?
Standard hot-pressed BN contains a small amount of boric oxide which is hygroscopic (absorbs moisture). If a substrate has been stored in a humid environment, it should be “baked out” at 100-150°C before being used in high-temperature chemical processes to prevent cracking from steam expansion.
4. Can you create custom shapes like threaded holes in BN substrates?
Yes. One of the primary benefits of Boron Nitride is its ability to be machined with complex features like internal/external threads, deep blind holes. And intricate channels for microfluidic chemical applications.
5. What is the maximum size for a BN ceramic substrate?
Size is generally limited by the size of the hot-pressed billet. At Great Ceramic, we can produce substrates up to approximately 300mm x 300mm, depending on the thickness and grade required.
Conclusion
The use of boron nitride ceramic substrate for chemical applications represents the pinnacle of material science in corrosive and high-thermal environments. Its unique non-wetting characteristics, combined with the ability to be precision-machined into complex geometries, makes it the material of choice for the next generation of chemical sensors, semiconductor tools. And metallurgical components.
Selecting the right grade of Boron Nitride and ensuring precise tolerances requires a partner with deep technical expertise. Great Ceramic provides the manufacturing rigor and engineering support necessary to transform raw BN into high-performance components that drive innovation in the chemical industry.
Contacter Great Ceramic pour une demande personnalisée usinage de la céramique des solutions adaptées à votre application.
“`
boron nitride ceramic substrate for chemical is widely used in advanced ceramic applications.