Alumina Ceramic for Medical Industry: Complete Technical Guide
The demand for high-performance biomaterials has positioned the глинозем ceramic for medical industry applications as a critical cornerstone in modern healthcare engineering. Facing stringent physiological environments, medical device engineers and orthopedic designers require materials that offer absolute bio-inertness, zero cytotoxicity. And exceptional wear resistance. Traditional metals like 316L stainless steel or titanium alloys often suffer from long-term degradation, releasing metallic ions that cause metallosis or inflammatory responses. By transitioning to high-purity medical-grade глинозем (Al₂O₃), engineers can achieve friction coefficients below 0.1 in articulating joints and maintain structural integrity in highly corrosive in-vivo conditions. This technical guide explores the material science, comparative metrics. And manufacturing protocols required to process medical alumina. For procurement managers and R&D teams seeking solutions, Great Ceramic provides прецизионная обработка керамики with ultra-tight tolerances down to ±0.005mm, ensuring your medical components meet flawless specifications. Need an immediate quote? Contact Great Ceramic’s engineering team today to discuss your medical device RFQ.
Свойства материала
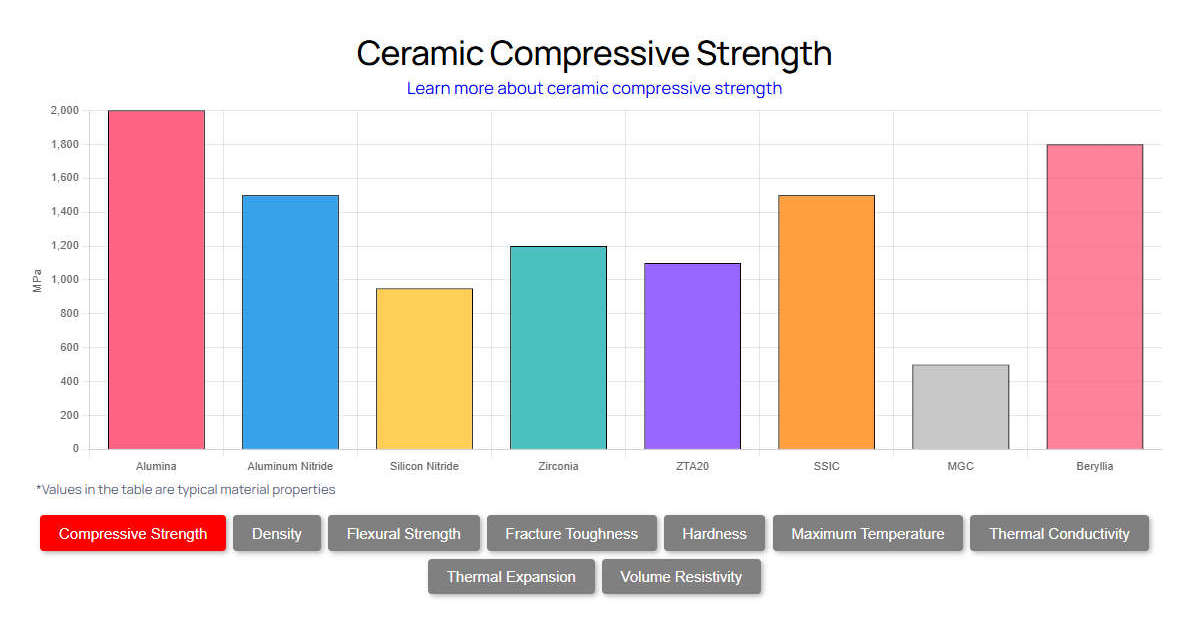
In medical applications, technical ceramics must conform to strict standards such as ASTM F603. This specifies high-purity dense aluminum oxide for surgical implant applications. Medical-grade alumina typically requires a purity of 99.5% to 99.9% to eliminate silica or alkali impurities that could compromise bio-stability. The resulting microstructure exhibits an average grain size of less than 4.5 µm. This maximizes mechanical strength and significantly reduces the probability of micro-crack propagation under cyclical physiological loading. The table below outlines the exact technical parameters of 99.9% high-purity medical alumina.
| Недвижимость | Значение | Единица |
|---|---|---|
| Плотность | 3.98 | г/см³ |
| Твердость | 2000 | HV |
| Прочность на изгиб | 400 | МПа |
| Вязкость разрушения | 4.5 | МПа-м½ |
| Теплопроводность | 35 | Вт/м-К |
| Электрическое сопротивление | >10¹⁴ | Ω-см |
| Максимальная рабочая температура | 1700 | °C |
Analyzing these parameters reveals why this material dominates the orthopedic and surgical sectors. The theoretical density of 3.98 g/cm³ ensures zero porosity. This is vital because surface pores can harbor bacteria or create stress concentrations leading to catastrophic failure under a 3,000 N physiological load. The extreme hardness of 2000 HV (Vickers) translates to negligible volumetric wear, typically measuring less than 0.01 mm³ per million articulation cycles in a simulated hip joint. Furthermore, the dielectric strength, indicated by an electrical resistivity exceeding 10¹⁴ Ω·cm, makes it an ideal insulator for high-frequency electrosurgical instruments operating at 300 kHz to 3 MHz, where current leakage could cause unintended tissue necrosis.
Сравнение с другими видами керамики
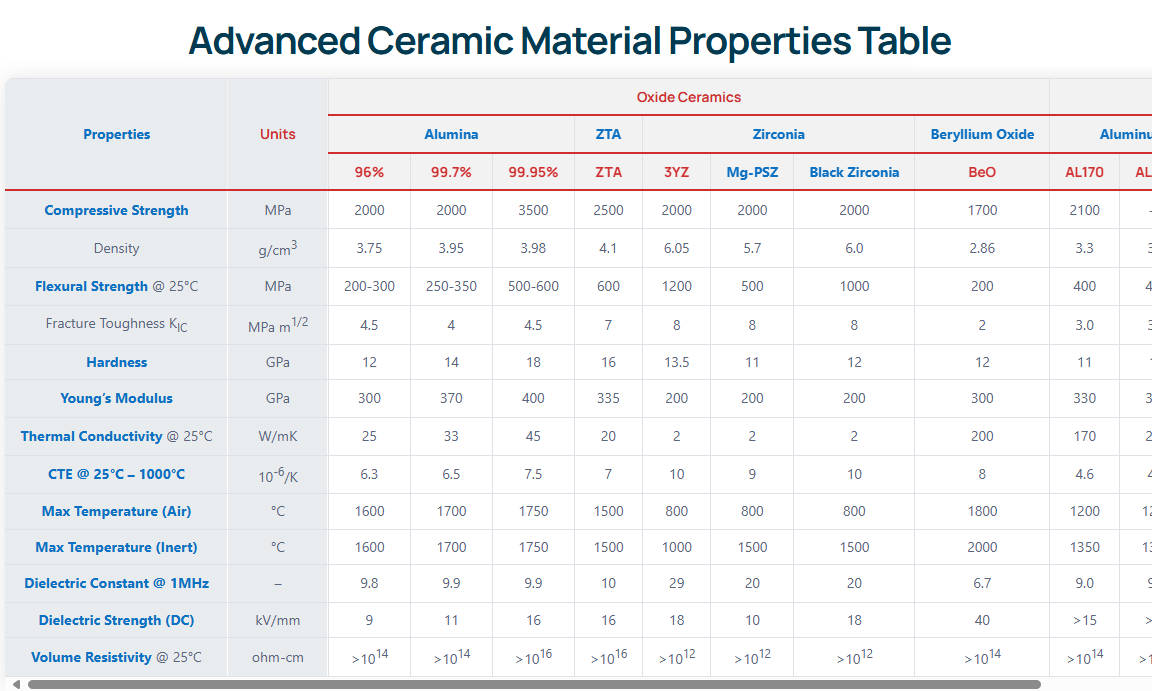
While alumina is the benchmark, engineers frequently evaluate it against other advanced ceramics like Yttria-Tetragonal Zirconia Polycrystal (Y-TZP) and нитрид кремния. Each material possesses distinct crystallographic behaviors under stress. The table below benchmarks the alumina ceramic for medical industry applications against standard industrial alumina and competing biomaterials.
| Недвижимость | Medical Alumina (99.9%) | Standard Alumina (95%) | Цирконий (Y-TZP) | Нитрид кремния |
|---|---|---|---|---|
| Теплопроводность | 35 | 24 | 2.5 | 30 |
| Твердость | 2000 | 1500 | 1200 | 1500 |
| Вязкость разрушения | 4.5 | 3.5 | 8.0 - 10.0 | 6.0 – 8.0 |
| Стоимость | Высокий | Низкий | Высокий | Очень высокий |
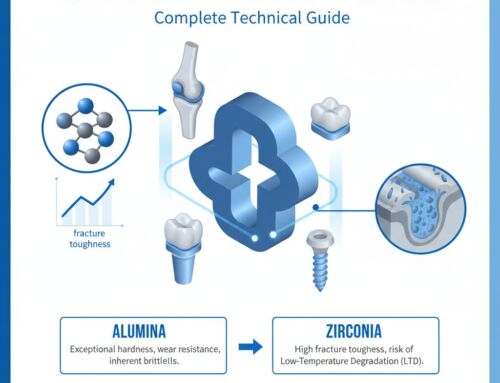
When engineering load-bearing implants, the primary trade-off exists between hardness (wear resistance) and fracture toughness (impact resistance). Medical alumina provides the highest hardness (2000 HV), making it superior for scratch resistance and articulation against Ultra-High-Molecular-Weight Polyethylene (UHMWPE). In contrast, диоксид циркония offers a higher fracture toughness (up to 10.0 MPa·m½) due to its phase-transformation toughening mechanism, where the tetragonal phase transforms into the monoclinic phase under stress, accompanied by a 4% volume expansion that naturally compresses advancing crack tips. However, pure zirconia is susceptible to Low-Temperature Degradation (LTD) in aqueous in-vivo environments over 10-15 years, a phenomenon strictly absent in alumina. Silicon nitride provides excellent biomechanical balance and inherent anti-bacterial properties but comes at a significantly higher raw material and processing cost. Standard 95% alumina is entirely unsuitable for long-term implantation due to its 5% glassy silicate phase. This dissolves in bodily fluids and drastically reduces flexural strength from 400 MPa down to less than 250 MPa over a 5-year period.
Приложения
- Orthopedic Joint Replacements (Total Hip Arthroplasty): Used primarily for femoral heads and acetabular cup inserts. The ±0.005mm sphericity achieved during final polishing ensures a perfect fluid-film lubrication regime. Engineers choose medical alumina here because its extreme hardness (2000 HV) and wettability result in a wear rate 100 times lower than Cobalt-Chrome (CoCr) alloy implants, virtually eliminating the risk of osteolysis caused by wear debris in younger, active patients.
- Dental Implants and Abutments: Employed in the maxiofacial sector for aesthetic, metal-free dental restorations. The bright white coloration mimics natural dentin, while its bio-inertness allows for superior osseointegration and soft-tissue attachment compared to titanium. With a flexural strength of 400 MPa, it easily withstands average human bite forces which range from 200 N to 700 N, ensuring long-term occlusal stability.
- Electrosurgical Instrument Insulation: Applied in minimally invasive laparoscopic and bipolar cautery tools. Operating at voltages up to 3,000 V-peak, these tools require flawless dielectric barriers. Alumina’s volume resistivity (>10¹⁴ Ω·cm) prevents catastrophic electrical arcing to non-target tissues. Its thermal stability allows the components to endure repeated steam autoclaving at 134°C and 2.1 bar pressure without hydrolytic degradation.
- Implantable Sensor Housings and Pacemaker Feedthroughs: Utilized to encapsulate sensitive microelectronics like cochlear implants and cardiac pacemakers. The material provides a hermetic seal when brazed with biocompatible titanium or platinum alloys. Its zero-porosity (density 3.98 g/cm³) blocks the ingress of corrosive interstitial fluids, protecting the electronics for device lifespans exceeding 15 years, while remaining perfectly transparent to RF telemetry signals for data transmission.
- Blood Pump Bearings and Valve Components: Integrated into Left Ventricular Assist Devices (LVADs) and mechanical heart valves. The high surface finish (Ra < 0.02 µm) prevents the adhesion of platelets, minimizing the risk of thrombosis (blood clotting). The low friction coefficient and dimensional stability under continuous flow (operating at 5,000 to 10,000 RPM) guarantee reliable hydrodynamic suspension without particulate shedding into the bloodstream.
Производственный процесс
The fabrication of high-purity alumina ceramic for medical industry applications demands an uncompromisingly clean environment to prevent metallic or organic contamination. The process begins with the synthesis of sub-micron, high-purity α-alumina powder (particle size 0.3 – 0.5 µm). To ensure uniform compaction, the powder is spray-dried with organic binders to form free-flowing spherical granules measuring 50 to 100 µm in diameter. The transition from raw powder to a ±0.005mm precision medical component requires absolute mastery over thermodynamics and CNC kinematics.
Методы формовки
- Холодное изостатическое прессование (CIP): For uniform, multi-directional stress distribution, the granulated powder is sealed in an elastomeric mold and subjected to hydrostatic liquid pressures between 200 MPa and 300 MPa. This method yields a highly uniform green body with a green density of approximately 60%, drastically reducing the risk of differential shrinkage and internal void formation during the firing stage.
- Керамическое литье под давлением (CIM): For complex, high-volume geometries like orthodontic brackets or surgical tool tips, the powder is compounded with a thermoplastic binder system (up to 40% volume fraction). The feedstock is injected into temperature-controlled molds under pressures of 50 to 100 MPa. The green parts then undergo a rigorous solvent and thermal debinding phase over 48 to 72 hours to remove the organics without inducing micro-fractures.
Спекание
Sintering is the thermodynamic process where the compacted green body coalesces into a dense ceramic. For medical-grade alumina, sintering is conducted in highly oxygenated or pure air atmospheres to prevent the reduction of Al₂O₃. The temperature ramp rate must be tightly controlled (typically 1°C to 2°C per minute) up to a peak temperature of 1600°C to 1650°C. The material is held at the peak temperature for 2 to 4 hours to achieve maximum densification (>99.5% theoretical density) while strictly suppressing abnormal grain growth. During this phase, the component undergoes a volumetric shrinkage of 15% to 20%. Accurately predicting this shrinkage is paramount to leaving the optimal amount of stock material (usually 0.2mm to 0.5mm) for the final hard machining operations.
Окончательная обработка
Due to the post-sintered hardness of 2000 HV, conventional high-speed steel or carbide tooling cannot cut alumina. Final machining must be executed using resin-bonded or metal-bonded diamond abrasives. To achieve the ±0.005mm tolerances required for medical devices, operations such as 5-axis ultrasonic-assisted grinding, cylindrical grinding. And precision lapping are utilized. Spindle speeds exceeding 20,000 RPM and fine diamond grits (D15 to D46) are employed under high-pressure water-based coolants (up to 70 bar) to evacuate swarf and prevent thermal shock at the cutting zone. Final surface polishing often utilizes diamond paste (1 µm to 0.25 µm particle sizes) on synthetic pitch laps to achieve an Ra finish of 0.02 µm, a mandatory specification for articulating joint surfaces.
Преимущества и ограничения
Преимущества
- Absolute Biocompatibility: Completely inert in the human body, meeting all ISO 10993 requirements for cytotoxicity, systemic toxicity. And sensitization, preventing the immunogenic responses commonly associated with metallic ions.
- Exceptional Wear Resistance: The extreme hardness (20 GPa) minimizes surface abrasion. In artificial hips, alumina-on-alumina bearings exhibit a wear rate of less than 1 mm³ after 20 years of simulated physiological use, outperforming CoCr and UHMWPE combinations by orders of magnitude.
- Chemical Inertness: Unaffected by harsh in-vivo enzymatic environments, blood pH variations (pH 7.35 – 7.45), or highly aggressive chemical sterilization agents (e.g., hydrogen peroxide plasma or ethylene oxide).
- High Compressive Strength: Withstands massive compressive loads exceeding 2500 MPa, allowing it to safely absorb peak biomechanical forces generated during activities like running or jumping without structural yielding.
Ограничения
- Inherent Brittleness: The relatively low fracture toughness (4.5 MPa·m½) makes the material highly sensitive to tensile stresses and impact loading. Design engineers must strictly avoid sharp internal radii, utilizing generous fillets (minimum R0.5mm) to mitigate stress concentration factors (Kt).
- Cost and Machining Difficulty: Processing alumina to tight tolerances after sintering requires costly diamond abrasive technology and extended machine hours. The hard-machining phase can account for up to 60% to 70% of the final component cost.
Особенности обработки
Transforming sintered alumina into a finished medical component requires navigating severe machining challenges. The primary difficulty stems from the material’s brittleness. improper feed rates or excessive depth of cut (DOC) leads to Sub-Surface Damage (SSD). This manifests as micro-cracks penetrating 10 µm to 50 µm below the machined surface. In a medical implant, these micro-cracks serve as initiation sites for catastrophic fatigue failure under dynamic biological loads. To mitigate this, material removal rates (MRR) must be kept strictly within the ductile-regime machining parameters, typically utilizing a DOC of less than 0.005 mm per pass during the final finishing stages. Furthermore, the selection of the grinding wheel matrix is critical. friable resin-bond diamond wheels are often preferred for final grinding as they continuously self-sharpen, maintaining low grinding forces (under 50 N) and preventing thermal degradation of the ceramic matrix.
Great Ceramic excels in overcoming these distinct challenges. Utilizing advanced multi-axis CNC grinding centers equipped with in-process laser metrology, our engineers maintain absolute control over the tool-path kinematics. The table below outlines our specific high-precision machining tolerances achievable for medical-grade alumina components.
| Machining Parameter / Feature | Great Ceramic Capability | Medical Industry Standard Requirement |
|---|---|---|
| Dimensional Tolerance | ±0.005 mm | ±0.020 mm |
| Шероховатость поверхности (Ra) | 0.02 µm | 0,05 мкм |
| Concentricity / Runout | 0.005 mm | 0.015 mm |
| Sphericity (Joint Heads) | 0.003 mm | 0.010 mm |
| Hole Drilling (Min. Diameter) | 0.150 mm | 0.500 mm |
Whether you require complex geometrical features for endoscopic tips or flawless spherical geometry for prosthetic articulation, precision is non-negotiable. Our strict quality control processes adhere to global medical manufacturing standards, ensuring every batch delivered is free from critical flaws. In addition to alumina, Great Ceramic applies these same exact tolerances to нитрид алюминия и карбид кремния components for advanced medical thermal management systems. Optimize your supply chain today—send your CAD files to Great Ceramic’s engineering team for a comprehensive Design for Manufacturability (DFM) review and accurate RFQ.
FAQ
What is alumina ceramic for medical industry?
The alumina ceramic for medical industry is a high-purity (99.5% to 99.9% Al₂O₃) advanced technical ceramic synthesized specifically for healthcare applications. Unlike industrial-grade ceramics, medical alumina is manufactured under strict clean-room conditions to eliminate silica, magnesia, or heavy metal impurities. It boasts a density of 3.98 g/cm³, zero porosity. And a hardness of 2000 HV. Because it is highly bio-inert and chemically stable, the human immune system does not recognize it as a foreign threat, preventing fibrous encapsulation or rejection. It is strictly regulated by international standards like ISO 6474-1 and ASTM F603, ensuring that the material is safe for permanent or temporary human implantation.
What are the main applications of alumina ceramic for medical industry?
The primary applications revolve around load-bearing and wear-resistant scenarios inside the human body. The most critical application is in Total Hip Arthroplasty (THA), where alumina is used to manufacture the femoral head and acetabular liner, drastically reducing joint wear rates to under 0.01 mm³ per million cycles. It is also heavily utilized in the dental sector for root implants and aesthetic crowns due to its tooth-like coloration and 400 MPa flexural strength. In active medical devices, alumina serves as the hermetic insulating feedthrough for cardiac pacemakers and cochlear implants, utilizing its >10¹⁴ Ω·cm electrical resistivity to protect microelectronics. Furthermore, its ability to withstand 134°C steam autoclaving makes it the material of choice for reusable surgical tool components, scalpel handles. And minimally invasive trocars.
How does alumina ceramic for medical industry compare to other ceramics?
When evaluated against other bioceramics, alumina stands out for its unmatched hardness and lowest wear rate. Compared to zirconia (Y-TZP). This has a higher fracture toughness of 8.0 – 10.0 MPa·m½ and higher flexural strength (up to 1000 MPa), alumina is slightly more brittle (4.5 MPa·m½). However, alumina is entirely immune to Low-Temperature Degradation (LTD), a process where zirconia slowly loses structural integrity over 10 to 15 years when exposed to warm bodily fluids. Compared to silicon nitride, alumina is less expensive to manufacture and has a longer, more proven clinical history in orthopedics, though silicon nitride offers unique anti-bacterial properties. Compared to industrial 95% alumina, medical 99.9% alumina contains no glassy intergranular phases, meaning it will not dissolve or weaken over time when exposed to the corrosive pH variations of human blood and interstitial fluid.
What are the advantages of alumina ceramic for medical industry?
The foremost advantage is its absolute biocompatibility. it elicits zero cytotoxic, mutagenic, or systemic toxic responses, making it entirely safe for lifelong implantation. Secondly, its ultra-high hardness (20 GPa) provides extreme wear resistance. This eliminates the generation of wear debris—a major cause of implant failure and bone loss (osteolysis) seen in metallic implants. Thirdly, its chemical inertness means it does not corrode or release ions, regardless of changes in bodily pH or prolonged exposure to aggressive sterilization chemicals. Fourthly, it possesses excellent compressive strength (over 2500 MPa), allowing it to easily support the mechanical loads of the human skeletal system. Finally, its excellent dielectric properties ensure safety in electrosurgical devices by preventing stray electrical currents from damaging patient tissue.
How is alumina ceramic for medical industry machined?
Machining medical alumina requires specialized hard-machining techniques because its 2000 HV hardness destroys traditional high-speed steel or carbide tooling instantly. After the raw powder is pressed and sintered at 1650°C, it shrinks by 15% to 20% and achieves its maximum hardness. The final precise dimensions are achieved using resin or metal-bonded diamond grinding wheels, rotating at high speeds (often exceeding 30 m/s cutting speeds). Copious amounts of high-pressure coolant (50-70 bar) are required to flush away ceramic swarf and control frictional heat. To prevent dangerous sub-surface micro-cracks, the depth of cut is tightly controlled to within a few microns per pass. Great Ceramic specializes in this exact rigorous process, utilizing state-of-the-art 5-axis CNC grinding and ultrasonic machining centers to achieve precise tolerances of ±0.005 mm, perfectly spherical joint heads (±0.003 mm). And mirror-like surface finishes of Ra 0.02 µm. Our engineering team ensures every component perfectly aligns with strict medical device specifications, optimizing both performance and patient safety.
Need custom alumina ceramic for medical industry parts? Связаться с компанией Great Ceramic для получения услуг по прецизионной обработке с жесткими допусками, или отправьте сообщение по электронной почте [email protected].
alumina ceramic for medical industry is widely used in advanced ceramic applications.
Узнайте больше о Alumina Ceramic For Medical Industry и наши услуги по прецизионной обработке керамики.