Aluminum Nitride Ceramic for Chemical Industry: Complete Technical Guide
The chemical processing sector operates under some of the most aggressive environments known to modern engineering, where components are routinely subjected to highly corrosive media, massive thermal gradients. And extreme pressures. Standard metallic alloys—even advanced superalloys like Hastelloy or Inconel—frequently succumb to pitting, oxidation. And catastrophic thermal fatigue when exposed to halogenated gases or rapid exothermic reactions. This is where nitrure d'aluminium ceramic for chemical industry applications becomes a critical engineering solution. Offering an exceptional thermal conductivity of up to 230 W/m·K combined with outstanding resistance to plasma, molten metals. And aggressive chemical reagents, nitrure d'aluminium (AlN) bridges the gap between thermal management and chemical inertness. For engineers and procurement managers seeking to eliminate localized hot spots in reactors or extend the Mean Time Between Failures (MTBF) in semiconductor chemical vapor deposition (CVD) equipment, specifying precision-machined AlN is paramount. At Great Ceramic, our usinage de précision de la céramique capabilities ensure that these advanced materials are delivered to exacting ±0.005mm tolerances, ready to integrate into your most demanding chemical infrastructure. Ready to optimize your reactor components? Submit an RFQ to Great Ceramic today to discuss your technical requirements.
Propriétés des matériaux
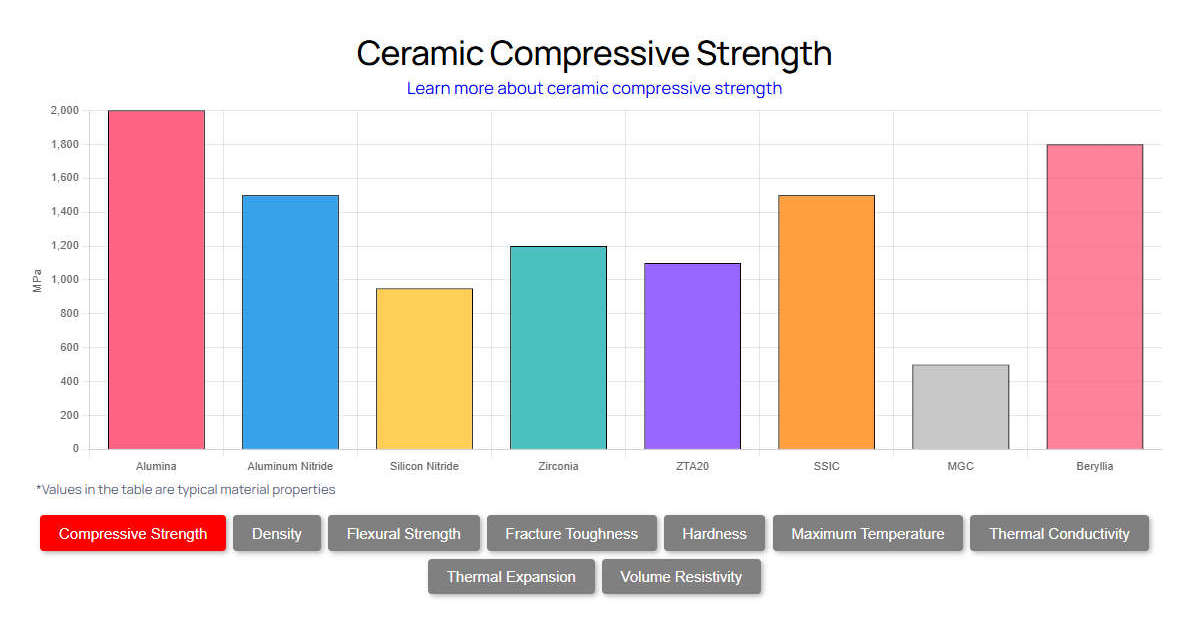
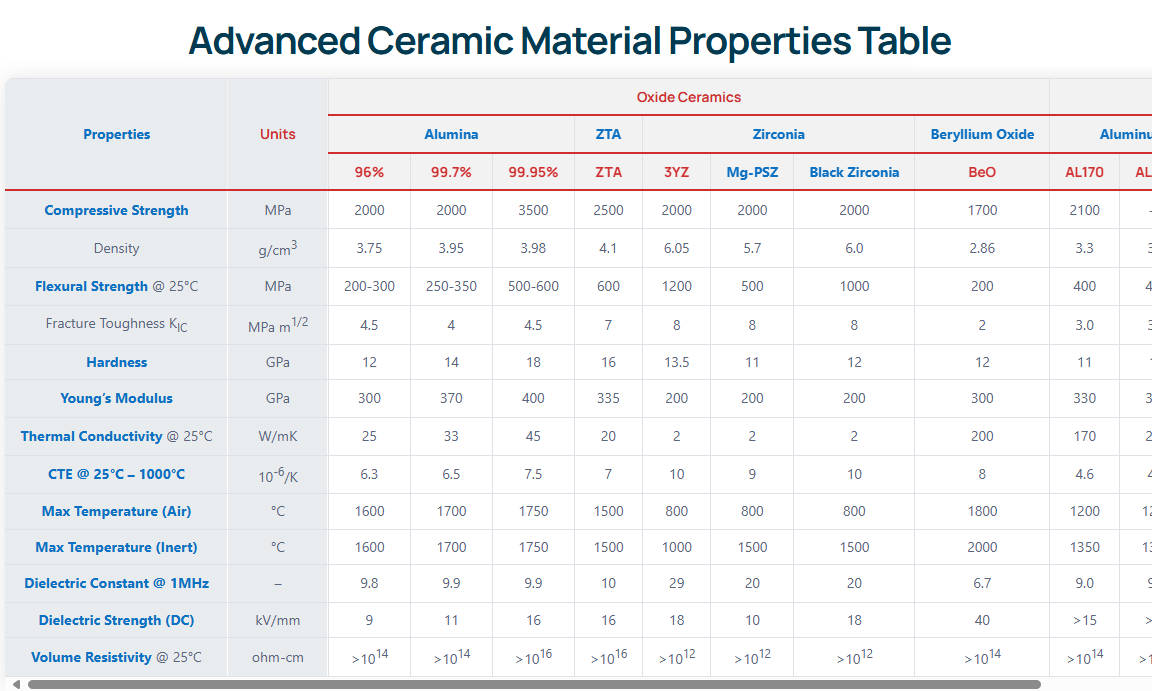
To fully understand why engineers specify aluminum nitride ceramic for chemical industry applications, one must examine its thermomechanical and electrical baseline. Unlike traditional oxides, AlN is a covalently bonded non-oxide ceramic. This unique atomic structure allows phonons to propagate with minimal scattering, resulting in a thermal conductivity that rivals metallic aluminum (237 W/m·K) while retaining the electrical insulation and chemical inertness of advanced technical ceramics. The theoretical maximum thermal conductivity of a perfect AlN single crystal is approximately 320 W/m·K, but commercially available sintered bodies range from 170 to 230 W/m·K depending on the yttria (Y₂O₃) sintering aids used to trap oxygen impurities.
Furthermore, its coefficient of thermal expansion (CTE) is exceptionally low at 4.5 × 10⁻⁶/°C (measured from 20°C to 400°C). This is nearly perfectly matched to that of silicon (4.0 × 10⁻⁶/°C). This makes it indispensable for hybrid chemical sensors and thermographic equipment. The material also exhibits excellent dielectric strength, routinely exceeding 15 kV/mm. This prevents electrical arcing in high-voltage electro-chemical processing cells. Below is the strict quantitative data defining commercial-grade AlN.
| Propriété | Valeur | Unité |
|---|---|---|
| Densité | 3.26 - 3.30 | g/cm³ |
| Dureté | 1100 - 1200 | HV |
| Résistance à la flexion | 300 - 340 | MPa |
| Résistance à la rupture | 2.8 - 3.5 | MPa-m½ |
| Conductivité thermique | 170 - 230 | W/m-K |
| Résistivité électrique | > 10¹⁴ | Ω-cm |
| Température maximale de fonctionnement | 1100 (Oxidizing) / 1900 (Inert) | °C |
Comparaison avec d'autres céramiques
Material selection in chemical engineering requires a rigorous cost-to-performance analysis. When evaluating aluminum nitride ceramic for chemical industry usage, it is critical to benchmark it against other leading industrial ceramics: Alumina (Al₂O₃), Zirconia (ZrO₂). And Silicon Nitride (Si₃N₄). Each material possesses a distinct thermomechanical profile.
alumine/”>Alumina is the industry workhorse, offering excellent baseline chemical resistance at a low cost, but its thermal conductivity peaks at merely 35 W/m·K. This makes Alumina highly susceptible to thermal shock in reactors with rapid exothermic temperature spikes. Zircone provides unmatched fracture toughness (up to 8.0 MPa·m½) and wear resistance, but it acts as a thermal insulator (2-3 W/m·K). This is disastrous for heat-dissipating chemical components. Nitrure de silicium offers superior mechanical strength and thermal shock resistance due to its interlocking needle-like grain structure, but its thermal conductivity (20-30 W/m·K) still falls drastically short of AlN. For chemical applications requiring simultaneous rapid heat dissipation and plasma erosion resistance—such as in fluorine-based etching chambers—AlN is the undisputed choice.
| Propriété | Nitrure d'aluminium (AlN) | Alumine (99.5%) | Zircone (Y-TZP) | Nitrure de silicium |
|---|---|---|---|---|
| Conductivité thermique (W/m-K) | 170 - 230 | 24 – 35 | 2 – 3 | 20 – 30 |
| Dureté (HV) | 1100 - 1200 | 1440 – 1500 | 1200 – 1300 | 1400 – 1600 |
| Résistance à la rupture (MPa-m½) | 2.8 - 3.5 | 4.0 - 5.0 | 6.0 – 8.0 | 6.0 - 7.0 |
| Coût | Haut | Faible | Moyen | Haut |
Applications
The integration of aluminum nitride ceramic for chemical industry infrastructure fundamentally alters the operational limits of processing equipment. By replacing legacy quartz, alumina, or coated metals with AlN, chemical engineers can operate at higher temperatures, utilize more aggressive reactive gases. And dramatically reduce cycle times through accelerated heating and cooling phases. The applications below highlight the critical intersection of AlN’s thermal management and chemical stability.
- Semiconductor Chemical Vapor Deposition (CVD) Susceptors: In epitaxial growth and CVD processes, silicon wafers must be heated uniformly to temperatures between 800°C and 1200°C while exposed to highly corrosive precursor gases like silane, ammonia. And hydrogen chloride. AlN susceptors provide a precise thermal profile (ΔT < 1°C across a 300mm wafer) due to their 170+ W/m·K thermal conductivity, while entirely resisting the halogenated plasma environments that would rapidly degrade lesser ceramics.
- Heat Exchanger Components in Corrosive Environments: Traditional shell-and-tube heat exchangers rely on stainless steel or Hastelloy. This suffer from severe pitting in the presence of hydrochloric (HCl) or hydrofluoric (HF) acids. Machined AlN tubes and plates offer heat transfer coefficients nearly identical to metallic aluminum, but are chemically inert to these aggressive acidic media, extending component lifespan from an average of 4,000 hours to over 20,000 hours of continuous operation.
- Crucibles for Molten Metal and Salt Processing: The extraction and refinement of rare earth elements often require molten salt electrolysis at temperatures exceeding 900°C. AlN is uniquely non-wetting to molten metals—particularly aluminum, lithium. And gallium. Crucibles machined from AlN do not contaminate the melt with oxygen impurities (unlike alumina crucibles) and can withstand the thermal shock of rapid induction heating cycles.
- High-Power UV-LED Substrates for Photochemical Reactors: Modern water treatment and chemical sterilization utilize high-intensity UV LEDs to break chemical bonds or destroy pathogens. These chips generate massive localized heat flux (often exceeding 100 W/cm²). AlN substrates pull this heat away from the semiconductor junction instantaneously, maintaining the diode’s junction temperature below 85°C, thereby preventing thermal degradation and ensuring stable photochemical reaction kinetics.
- Analytical Chemistry Instrumentation Sensors: Thermal Conductivity Detectors (TCD) used in gas chromatography rely on measuring the differential cooling of a heated filament in the presence of various carrier gases. Utilizing AlN sensor housings provides absolute baseline thermal stability, preventing external ambient temperature fluctuations from causing sensor drift, thereby increasing the precision of chemical mass analysis down to parts-per-billion (ppb) thresholds.
Processus de fabrication
Achieving the theoretical properties of aluminum nitride ceramic for chemical industry deployment requires an exceptionally controlled manufacturing pipeline. Because AlN is highly sensitive to oxygen contamination—which creates vacancies in the crystal lattice that scatter phonons and destroy thermal conductivity—the entire process, from powder synthesis to final machining, must be rigorously managed. The standard synthesis begins with carbothermal reduction, where high-purity alumina is reacted with carbon and nitrogen gas at 1600°C (Al₂O₃ + 3C + N₂ → 2AlN + 3CO). The resulting powder is sub-micron in size, highly reactive. And must be handled in inert atmospheres.
Méthodes de formage
- Coulée de bandes : For thin substrates used in high-power chemical sensors or LED heat sinks, the AlN powder is mixed with organic binders, plasticizers. And solvents to create a slurry. This slurry is cast via a doctor blade into precise green sheets ranging from 0.25mm to 1.5mm in thickness.
- Pressage isostatique à froid (CIP) : For volumetric components such as crucibles, heat exchanger blocks. And susceptors, the powder is placed in a flexible elastomeric mold and subjected to omnidirectional hydraulic pressure (typically 200 to 300 MPa). This ensures a uniform green density, preventing warping and anisotropic shrinkage during the sintering phase.
Frittage
Sintering AlN is vastly more complex than oxide ceramics. Because of its covalent bonding, AlN exhibits very low self-diffusion rates. It requires temperatures between 1700°C and 1900°C in a nitrogen atmosphere. Furthermore, liquid phase sintering is mandatory. Sintering aids, typically Yttria (Y₂O₃) or Calcium Oxide (CaO), are added at 3-5 wt%. During sintering, the Y₂O₃ reacts with the inherent oxygen layer (Al₂O₃) on the surface of the AlN particles to form yttrium aluminate liquid phases (e.g., Y₃Al₅O₁₂, known as YAG). This liquid phase not only densifies the ceramic to >99% theoretical density but essentially “sweeps” oxygen out of the AlN lattice, migrating it to the grain boundaries. This oxygen purification step is exactly what allows the final material to achieve thermal conductivities exceeding 170 W/m·K.
Usinage final
In its as-sintered state, AlN parts shrink by roughly 18-20% and exhibit a surface finish and dimensional tolerance inadequate for strict chemical engineering applications. Final machining is executed using state-of-the-art multi-axis CNC grinding centers. Because AlN possesses a hardness of 1100-1200 HV, traditional carbide tooling is instantly destroyed. Diamond-impregnated grinding wheels and ultra-precise lapping plates are utilized. At Great Ceramic, our engineers meticulously control the cutting depth, feed rate. And spindle speeds to prevent micro-cracking, achieving tight dimensional tolerances of ±0.005mm and surface roughness (Ra) down to 0.1μm.
Avantages et limites
While aluminum nitride ceramic for chemical industry uses provides a unique matrix of properties, it is not a universal panacea. Engineers must carefully weigh its specialized thermomechanical advantages against its environmental and mechanical limitations to ensure optimal deployment in chemical reactors and analytical hardware.
Avantages
- Unmatched Thermal Conductivity: At 170-230 W/m·K, AlN transfers heat 5 to 7 times faster than alumina, eliminating thermal gradients that cause differential expansion and catastrophic failure in chemical vessels.
- Exceptional Halogen Resistance: AlN demonstrates near-zero mass loss when exposed to fluorine gas, chlorine gas. And standard plasma etching environments at elevated temperatures, vastly outperforming quartz and borosilicate glass.
- Non-Wetting Characteristics: The material strongly resists wetting by molten metals—particularly Group III elements like aluminum and gallium—making it the premier choice for pyrometallurgical chemical extraction components.
- Matched Coefficient of Thermal Expansion (CTE): Its CTE of 4.5 × 10⁻⁶/°C closely mirrors silicon (4.0 × 10⁻⁶/°C), preventing delamination and mechanical stress in chemically bonded silicon-to-ceramic assemblies during rapid thermal cycling.
Limitations
- Susceptibility to High-Temperature Hydrolysis: AlN is generally stable in water at room temperature, but in aqueous chemical environments exceeding 80°C, the surface can react with water (hydrolysis) to form aluminum hydroxide and ammonia gas (AlN + 3H₂O → Al(OH)₃ + NH₃). It must be protected or avoided in high-temperature steam applications.
- High Production and Machining Costs: The requirement for ultra-pure powder synthesis, high-temperature nitrogen sintering. And diamond grinding makes AlN significantly more expensive than alumina or steatite ceramics, relegating its use to mission-critical components where failure is unacceptable.
Considérations relatives à l'usinage
The very characteristics that make aluminum nitride ceramic for chemical industry use so desirable—its covalent bonding, extreme hardness (1100+ HV). And rigidity—also make it notoriously difficult to machine. In the field of advanced ceramics, machinability is often inversely proportional to performance. AlN is highly brittle, possessing a fracture toughness of merely 2.8 to 3.5 MPa·m½. When subjected to the shear forces of CNC grinding, the material is prone to a phenomenon known as Sub-Surface Damage (SSD), where micro-cracks propagate below the machined surface. In chemical applications, these microscopic fissures can act as capillary networks, allowing corrosive gases to infiltrate and prematurely degrade the component.
To overcome these severe machining challenges, conventional milling and turning parameters must be entirely discarded. Great Ceramic utilizes specialized diamond-bonded abrasive tools operating at ultra-high spindle speeds (frequently exceeding 15,000 RPM) coupled with exceedingly low feed rates (down to 0.01 mm/rev). This kinematic approach ensures that material removal occurs in the “ductile regime”—a microscopic zone where the ceramic is ground away without initiating brittle fracture. Coolant management is another critical hurdle. because AlN can undergo hydrolysis, specialized non-aqueous or chemically inhibited cutting fluids must be flooded precisely into the cutting zone to evacuate swarf and manage friction-induced heat.
Edge chipping is a persistent threat when machining complex geometries like gas distribution showerheads or threaded fluid connectors for chemical reactors. Our technicians utilize customized chamfering protocols and sacrificial backing materials to eliminate breakout as the diamond tool exits the cut. By combining 5-axis CNC grinding technology with rigorous metrology (CMM inspection), Great Ceramic routinely supplies AlN components with complex internal fluidic channels, achieving planar flatness of 2 light bands and geometrical tolerances of ±0.005mm. If your engineering team is struggling to source high-precision AlN parts that meet chemical industry standards, consult our usinage de précision de la céramique experts to optimize your design for manufacturability.
FAQ
What is aluminum nitride ceramic for chemical industry applications?
Aluminum nitride (AlN) is an advanced, non-oxide technical ceramic covalently bonded from aluminum and nitrogen. In the chemical industry, it is utilized primarily for its rare combination of massive thermal conductivity (up to 230 W/m·K) and robust resistance to aggressive chemical attacks, particularly from halogens, plasmas. And molten metals. Unlike metallic alloys that corrode, or standard ceramics that suffer thermal shock, AlN provides a stable, pure platform for high-temperature chemical reactions, semiconductor processing. And thermal management systems.
What are the main applications of aluminum nitride in chemical processing?
The primary applications revolve around environments requiring extreme heat transfer and corrosion resistance simultaneously. Key uses include susceptors and heating elements for semiconductor Chemical Vapor Deposition (CVD), heat exchanger plates for highly acidic fluid processing, crucibles for melting rare earth metals and salts without oxygen contamination. And housings for sensitive analytical chemistry sensors (like gas chromatographs) where thermal stability is strictly required for accurate readings.
How does aluminum nitride compare to other ceramics like alumina or zirconia?
The most striking difference is thermal conductivity. AlN transfers heat at 170-230 W/m·K. This is nearly 7 times faster than standard Alumina (24-35 W/m·K) and almost 100 times faster than Zirconia (2-3 W/m·K). While Zirconia is far tougher (up to 8.0 MPa·m½) and Alumina is vastly more cost-effective for general chemical wear, neither can survive the severe thermal shock environments that AlN handles effortlessly. Additionally, AlN offers a much closer CTE match to silicon than both alumina and zirconia.
What are the advantages of using aluminum nitride over metals in chemical reactors?
Metals, including superalloys like Inconel or Hastelloy, degrade rapidly when exposed to halogens (fluorine, chlorine) at high temperatures, resulting in particulate contamination of the chemical batch. AlN is virtually inert to these gases. Furthermore, metals are electrically conductive. AlN offers similar thermal conductivity to aluminum metal but acts as a powerful electrical insulator (dielectric strength > 15 kV/mm), making it ideal for electro-chemical reactors, plasma chambers. And applications where electrical shorts must be strictly prevented.
How is aluminum nitride machined to tight tolerances?
Due to its hardness (over 1100 HV) and low fracture toughness (approx. 3.0 MPa·m½), AlN cannot be machined with standard high-speed steel or carbide tooling. It requires precision grinding using diamond-impregnated tools on rigid, vibration-dampened CNC platforms. The process demands exact control of feed rates, spindle RPM. And specialized coolants to prevent micro-cracking and hydrolysis during cutting. Great Ceramic utilizes advanced 5-axis grinding and lapping technology to machine AlN components to ultra-tight tolerances of ±0.005mm, ensuring flawless integration into critical chemical industry infrastructure.
Need custom aluminum nitride ceramic for chemical industry parts? Contacter Great Ceramic pour des services d'usinage de précision avec des tolérances serrées, ou envoyez un courriel à l'adresse suivante [email protected].
aluminum nitride ceramic for chemical industry is widely used in advanced ceramic applications.
En savoir plus sur Céramique de nitrure d'aluminium pour l'industrie chimique et nos services d'usinage céramique de précision.