Aluminum Nitride Ceramic Substrate for Chemical: Complete Technical Guide
The deployment of advanced technical ceramics in aggressive chemical environments presents a complex engineering challenge, particularly when high thermal dissipation and strict dimensional stability are required simultaneously. The aluminum nitride ceramic substrate for chemical applications has emerged as the premier solution for engineers dealing with highly exothermic reactions, corrosive gas environments. And plasma-based chemical vapor deposition (CVD) processes. Unlike traditional oxide ceramics, aluminum nitride (AlN) offers an exceptional theoretical thermal conductivity of up to 320 W/m·K (commercially available at 170-230 W/m·K), paired with outstanding resistance to halogenated gases and fluorine plasmas. This guide explores the thermomechanical properties, material comparisons. And specialized manufacturing processes required to produce these substrates. At Great Ceramic, we specialize in overcoming the inherent brittleness of AlN to deliver tight-tolerance substrates (±0.005mm) that ensure zero-leakage interfaces and optimal heat transfer for demanding chemical processing equipment. For immediate project evaluation, contact our engineering team.
Material Properties
Understanding the thermomechanical and chemical profile of the aluminum nitride crystal lattice is critical for specifying substrates in harsh environments. AlN features a wurtzite crystal structure with strong covalent bonds between aluminum and nitrogen atoms. This specific atomic arrangement facilitates highly efficient phonon-mediated heat transfer. While metals rely on electron mobility for thermal conductivity (which makes them electrically conductive), AlN relies on lattice vibrations (phonons), resulting in a substrate that is simultaneously an excellent thermal conductor and a superior electrical insulator with a dielectric strength exceeding 15 kV/mm.
In chemical applications, structural integrity under thermal stress is paramount. The material exhibits a coefficient of thermal expansion (CTE) of 4.5 x 10⁻⁶ /°C (measured between 20°C and 400°C). This closely matches silicon (4.0 x 10⁻⁶ /°C). This matching prevents delamination and microscopic thermal fatigue cracking during rapid exothermic chemical reactions or localized plasma heating. The data table below outlines the definitive properties of high-purity sintered AlN substrates utilized in chemical-grade engineering.
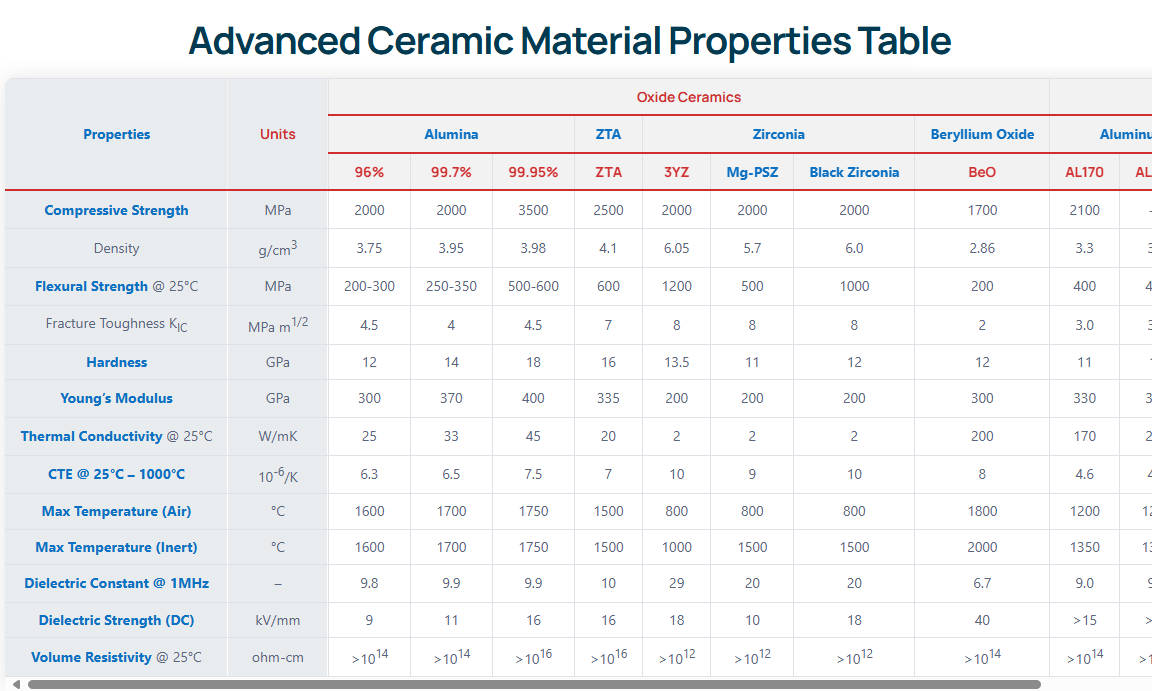
| Property | Value | Unit |
|---|---|---|
| Density | 3.26 – 3.30 | g/cm³ |
| Hardness | 1000 – 1100 | HV |
| Flexural Strength | 300 – 400 | MPa |
| Fracture Toughness | 2.6 – 3.5 | MPa·m½ |
| Thermal Conductivity | 170 – 230 | W/m·K |
| Electrical Resistivity | > 10¹⁴ | Ω·cm |
| Max Working Temperature | 1000 (Air) / 1900 (Inert) | °C |
The flexural strength of 300 to 400 MPa ensures that the substrate can withstand significant fluidic pressure and mechanical clamping forces inherent in chemical reactor assemblies. However, the fracture toughness of 2.6 to 3.5 MPa·m½ indicates moderate brittleness, necessitating precise machining to eliminate surface micro-cracks that could serve as failure initiation points under mechanical or thermal shock.
Comparison with Other Ceramics
Selecting the correct ceramic substrate requires a rigorous evaluation of competing technical ceramics. While AlN excels in specific domains, it must be cross-referenced against oxide and non-oxide alternatives to validate its selection for chemical infrastructure. The following table provides a direct numerical comparison across vital engineering metrics.
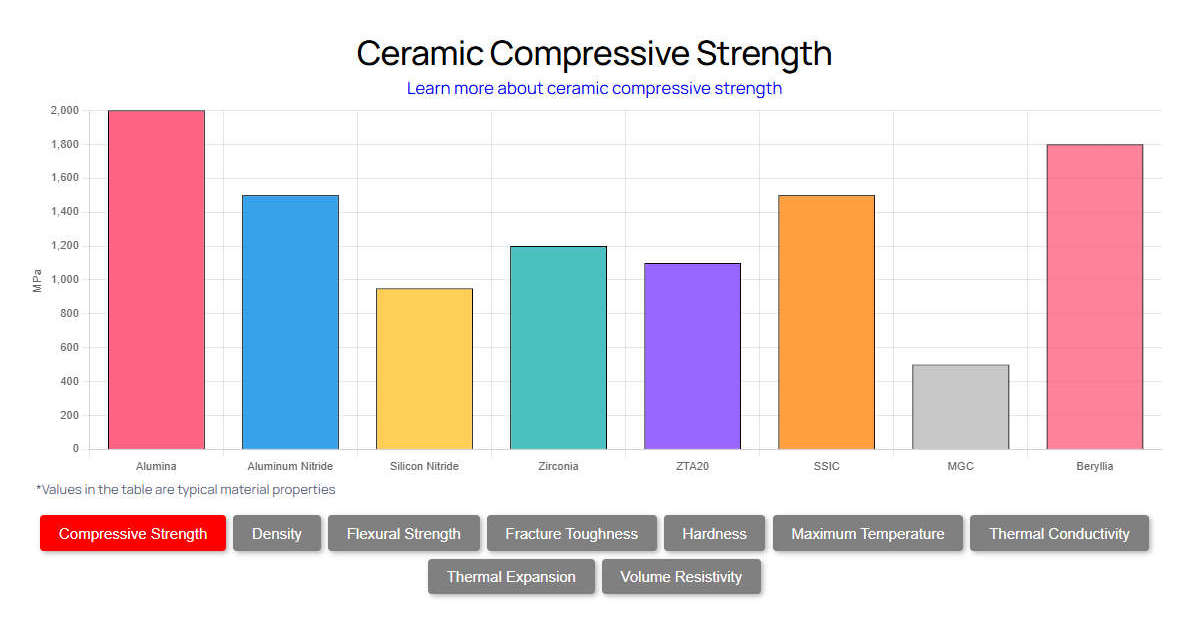
| Property | Aluminum Nitride | Alumina | Zirconia | Silicon Nitride |
|---|---|---|---|---|
| Thermal Conductivity (W/m·K) | 170 – 230 | 20 – 30 | 2 – 3 | 30 – 90 |
| Hardness (HV) | 1000 – 1100 | 1400 – 1600 | 1200 – 1300 | 1400 – 1700 |
| Fracture Toughness (MPa·m½) | 2.6 – 3.5 | 3.5 – 4.5 | 8.0 – 10.0 | 6.0 – 8.0 |
| Cost | High | Low | Medium | High |
When engineering chemical systems, the choice between AlN and standard alumina/”>alumina (Al₂O₃) often comes down to thermal management. Alumina is highly cost-effective and chemically inert, but its low thermal conductivity (20-30 W/m·K) makes it a thermal bottleneck in highly exothermic micro-reactor designs. If a reaction generates localized heat exceeding 50 W/cm², an alumina substrate will induce severe thermal gradients, potentially altering reaction kinetics or causing catastrophic substrate failure. In contrast, an aluminum nitride ceramic substrate for chemical applications quickly dissipates this heat, maintaining isothermal conditions across the reactor bed.
Compared to zirconia (ZrO₂). This boasts an exceptionally high fracture toughness (up to 10.0 MPa·m½), AlN is much more brittle. Zirconia is frequently used for chemical pump plungers and valves where impact resistance and wear are critical. However, zirconia acts as a thermal insulator (2-3 W/m·K). Therefore, for substrates requiring rapid heat extraction from chemical mixtures, zirconia is entirely unsuitable, leaving AlN as the necessary choice.
The closest non-oxide competitor is silicon nitride (Si₃N₄). Silicon nitride offers superior fracture toughness (6.0-8.0 MPa·m½) and excellent thermal shock resistance. However, in aggressive chemical vapor deposition environments—particularly those utilizing fluorine (F₂), nitrogen trifluoride (NF₃), or sulfur hexafluoride (SF₆) plasmas—AlN vastly outperforms Si₃N₄. The aluminum in AlN reacts with fluorine gas to form a microscopic passivation layer of aluminum fluoride (AlF₃), measuring only a few nanometers thick. This layer halts further etching. Silicon nitride, conversely, forms volatile silicon tetrafluoride (SiF₄) under the same conditions, leading to rapid material degradation and particle contamination in the chemical chamber.
Applications
The unique intersection of extreme thermal conductivity, specific dielectric properties. And chemical inertness to halogens makes the aluminum nitride ceramic substrate for chemical usage indispensable across multiple advanced industrial sectors. Below are the primary applications where engineers specify this material to solve complex system failures.
- Semiconductor Chemical Vapor Deposition (CVD) Heaters: In high-vacuum CVD and Etch chambers, substrate heaters must operate at temperatures up to 800°C while resisting aggressive cleaning plasmas (like NF₃ and CF₄). Aluminum nitride is chosen because its thermal conductivity allows for rapid, uniform heating (±1% temperature uniformity across a 300mm wafer), while its surface chemically resists plasma erosion far better than quartz or alumina, preventing particulate contamination in sub-3nm node chip manufacturing.
- Exothermic Chemical Micro-Reactors: Flow chemistry relies on micro-channel reactors to process highly exothermic, fast-kinetic reactions. AlN substrates are utilized to cap and encapsulate these micro-channels. The 170+ W/m·K thermal conductivity ensures that heat generated by reactions (such as organometallic syntheses or hazardous oxidations) is instantly wicked away to external cooling blocks, preventing thermal runaway and maximizing product yield.
- Analytical Chemistry Mass Spectrometry Insulators: Within the ion trap and source regions of advanced mass spectrometers, components are subjected to high voltage fields (up to 10 kV) and high temperatures, often in the presence of ionized carrier gases. AlN is chosen because its >10¹⁴ Ω·cm volume resistivity prevents electrical arcing, while its thermal capabilities maintain dimensionally stable alignments (due to its low 4.5 ppm/K CTE) between the critical focusing lenses.
- Petrochemical Sensor Substrates: Downhole drilling and petrochemical refining environments subject sensors to hydrogen sulfide (H₂S), high pressures (>20,000 psi). And temperatures exceeding 250°C. AlN substrates are used to mount the pressure and temperature sensor dies. The material is selected because it does not chemically degrade in acidic gas environments and transfers thermal data from the fluid to the sensor die with near-zero latency compared to standard encapsulation materials.
- High-Power UV-LED Curing Systems for Photochemistry: Industrial photochemical synthesis and chemical adhesive curing rely on dense arrays of ultra-violet (UV) LEDs. These arrays convert over 60% of their input power into heat. An aluminum nitride ceramic substrate for chemical curing applications acts as the direct sub-mount for the LED chips, rapidly moving heat away from the semiconductor junctions to prevent wavelength shift and premature diode failure.
Manufacturing Process
The production of an industrial-grade aluminum nitride ceramic substrate for chemical applications is a highly controlled, multi-stage metallurgical and ceramic engineering process. Because AlN possesses strong covalent bonds, it requires extremely high temperatures and specific chemical additives to achieve full densification. The journey from raw powder to a precision-machined substrate involves several critical phases.
Forming Methods
- Tape Casting (Doctor Blade Method): This is the dominant method for producing thin substrates (0.25mm to 2.0mm thick). High-purity AlN powder (typically with an average particle size of 1.0 to 3.0 μm) is mixed with organic binders, plasticizers. And non-aqueous solvents to create a viscous slurry. The slurry is cast under a precisely calibrated blade onto a moving polymer carrier tape. The solvents evaporate, leaving a flexible “green tape” that is subsequently cut into discrete substrate squares.
- Isostatic Pressing: For thicker chemical reactor components or custom 3D substrate profiles, dry pressing or cold isostatic pressing (CIP) is utilized. The powder is agglomerated into spray-dried granules, placed into a flexible mold. And subjected to hydraulic pressures exceeding 200 MPa to create a dense green body capable of being pre-machined before sintering.
Sintering
Sintering aluminum nitride is exceptionally challenging due to its low self-diffusion coefficient. To achieve >99% theoretical density, liquid phase sintering is employed. Sintering aids, primarily Yttrium Oxide (Y₂O₃) or Calcium Oxide (CaO), are added to the powder matrix at concentrations of 2% to 5% by weight. During the sintering process—which occurs in high-temperature graphite furnaces at 1800°C to 1950°C under a pure nitrogen atmosphere—the Y₂O₃ reacts with the native alumina layer on the surface of the AlN particles. This reaction forms yttrium aluminate liquid phases (such as Y₃Al₅O₁₂, or YAG) at the grain boundaries.
This liquid phase promotes densification and, crucially, sequesters oxygen impurities away from the AlN crystal lattice. Oxygen is the primary enemy of thermal conductivity in AlN. even 1% oxygen contamination can drop thermal conductivity from 200 W/m·K to below 100 W/m·K. The resulting sintered microstructure consists of pristine AlN grains surrounded by thin, continuous yttrium aluminate grain boundary phases.
Final Machining
The as-sintered substrates undergo rigorous post-processing to meet the geometric tolerances required for chemical sealing. Due to a post-sintering hardness of up to 1100 HV, traditional high-speed steel or carbide tooling is useless. Shaping the substrate requires specialized precision ceramic machining techniques. Substrates are ground flat using double-sided planetary lapping machines loaded with diamond abrasives. To achieve surface roughness values of Ra 0.05 μm—necessary for vacuum-tight seals in chemical CVD equipment—successive stages of fine diamond polishing paste (down to 0.5 μm grit) are applied under heavy pressure. Through-holes and internal vias for chemical flow are generated using ultrasonic core drilling or CNC laser ablation.
Advantages & Limitations
Implementing an aluminum nitride ceramic substrate for chemical engineering systems requires a balanced understanding of its intrinsic strengths and inherent operational weaknesses. Material selection is always a compromise. And recognizing these boundaries is essential for successful system integration.
Advantages
- Unmatched Thermal Management: With thermal conductivity ranging from 170 to 230 W/m·K, AlN exceeds the thermal capabilities of metallic aluminum (approx. 210 W/m·K) while remaining electrically insulating. This allows for direct metallization and direct component mounting without intervening thermal interface materials (TIMs) that introduce thermal resistance.
- Fluorine Plasma Immunity: In semiconductor chemistry, AlN exhibits near-zero etch rates in NF₃, CF₄. And SF₆ plasmas. The formation of the AlF₃ passivation layer ensures that critical tolerances are maintained over tens of thousands of process hours, drastically reducing equipment downtime and particulate generation.
- Silicon-Matched CTE: The CTE of 4.5 ppm/K aligns perfectly with silicon (4.0 ppm/K) and closely with gallium arsenide (GaAs) and silicon carbide (SiC) semiconductors. When these materials are brazed or soldered to the AlN substrate, the assembly experiences minimal bi-metallic thermal bowing during temperature cycling from -50°C to +200°C.
- High Dielectric Strength: A breakdown voltage exceeding 15 kV/mm makes AlN ideal for separating high-voltage components from cooling fluids in electrophoretic chemical separators and high-power laser diode drivers.
Limitations
- Vulnerability to Hydrolysis: Bare, unmetallized AlN is susceptible to chemical attack by water (hydrolysis) at elevated temperatures or in highly alkaline solutions. The reaction produces aluminum hydroxide and ammonia gas (AlN + 3H₂O → Al(OH)₃ + NH₃). This degrades the surface finish and structural integrity. In applications involving aqueous chemistry, the AlN substrate must be protected with passivating coatings (such as glassy silica layers) or thick-film metallization.
- High Cost and Processing Complexity: The requirement for ultra-high purity starting powders, rare-earth sintering aids, 1900°C nitrogen-atmosphere furnaces. And extensive diamond machining drives the cost of AlN significantly higher than alumina. It should be reserved for applications where its specific thermal and halogen-resistant properties are non-negotiable.
Machining Considerations
The transition from a raw sintered blank to a finished aluminum nitride ceramic substrate for chemical applications demands exceptional metrology and machining expertise. The primary challenge in machining AlN lies in managing brittle fracture mechanics. Unlike metals. This undergo plastic deformation when cut, ceramics remove material through controlled micro-fracturing. If machining forces (feed rates, spindle speeds, or depth of cut) exceed the localized fracture toughness of the substrate, macroscopic cracking or sub-surface damage occurs.
For chemical applications, surface integrity is just as critical as dimensional accuracy. Sub-surface micro-cracks act as initiation sites for chemical attack or thermal shock failure. At Great Ceramic, our engineering team mitigates these risks by utilizing high-frequency ultrasonic machining and multi-axis CNC grinding centers equipped with resin-bonded diamond wheels. Furthermore, because AlN is prone to hydrolysis, we strictly control the chemistry of our cutting fluids, utilizing specialized non-aqueous coolants or pH-buffered solutions that immediately remove grinding swarf without initiating ammonia off-gassing.
Below is a summary of the tight-tolerance machining capabilities we routinely achieve for custom chemical AlN substrates.
| Geometric Feature | Standard Tolerance | Advanced Capability | Metrology Method |
|---|---|---|---|
| Thickness | ±0.010 mm | ±0.005 mm | Laser Micrometer |
| Flatness (per 100mm) | 0.005 mm | 0.002 mm | Optical Interferometer |
| Hole Diameter | ±0.020 mm | ±0.010 mm | Coordinate Measuring Machine (CMM) |
| Surface Roughness (Ra) | 0.4 – 0.8 μm | 0.05 – 0.1 μm | Stylus Profilometer |
Achieving a flatness of 0.002 mm and a surface finish of Ra 0.05 μm is particularly critical for high-vacuum chemical CVD heaters, where the mating surfaces must achieve a hermetic seal against reactive gases without the use of elastomer O-rings. This would melt at high temperatures. By strictly controlling wheel grit degradation and managing thermal buildup during the grinding process, Great Ceramic ensures every substrate delivered is ready for immediate integration into severe environments.
FAQ
What is an aluminum nitride ceramic substrate for chemical applications?
An aluminum nitride (AlN) ceramic substrate is an advanced technical component made from highly densified AlN powder. In chemical applications, it serves as a foundational platform for mounting sensors, heaters, or micro-reactors. It is specifically engineered to provide extremely high thermal conductivity (up to 230 W/m·K) for rapid heat dissipation, excellent electrical insulation. And superior chemical resistance to aggressive environments, particularly halogenated gases and fluorine plasmas used in semiconductor processing.
What are the main applications of this substrate?
The primary applications revolve around environments requiring simultaneous thermal management and chemical resistance. Key uses include substrate heaters in chemical vapor deposition (CVD) and physical vapor deposition (PVD) chambers, encapsulating plates for exothermic continuous-flow micro-reactors, mounting boards for high-power UV-LED chemical curing arrays. And insulating supports within high-temperature mass spectrometry analytical instruments.
How does an aluminum nitride substrate compare to other ceramics?
Compared to standard alumina (Al₂O₃), AlN offers nearly 10 times the thermal conductivity, making it far superior for heat dissipation. Compared to zirconia (ZrO₂), AlN is much more brittle but vastly outperforms zirconia thermally, as zirconia is essentially a thermal insulator. When evaluated against silicon nitride (Si₃N₄), AlN exhibits superior resistance to fluorine-based plasma etching, although silicon nitride possesses higher mechanical fracture toughness. Cost-wise, AlN is a premium material compared to alumina.
What are the specific chemical advantages of aluminum nitride?
The most significant chemical advantage of AlN is its immunity to dry halogen environments and plasma erosion. When exposed to fluorine or chlorine gases at elevated temperatures, AlN forms a stable, microscopic aluminum halide passivation layer that prevents further degradation. Additionally, its high thermal conductivity ensures that localized hot spots—which can unpredictably accelerate chemical corrosion rates on other substrates—are rapidly normalized.
How is this technical ceramic machined to tight tolerances?
Due to its hardness of over 1000 HV and inherent brittleness, AlN cannot be machined with standard metal-cutting tools. It requires precision ceramic machining using diamond-impregnated grinding wheels, ultrasonic core drilling. And multi-stage planetary lapping. To prevent hydrolysis during manufacturing, specialized non-aqueous coolants are used. Great Ceramic utilizes state-of-the-art CNC equipment to achieve exceptional precision, offering advanced capabilities such as ±0.005mm thickness tolerances and Ra 0.05 μm surface finishes to ensure hermetic chemical sealing.
Need custom aluminum nitride ceramic substrate for chemical parts? Contact Great Ceramic for precision machining services with tight tolerances, or email [email protected].
aluminum nitride ceramic substrate for chemical is widely used in advanced ceramic applications.
Learn more about Aluminum Nitride Ceramic Substrate For Chemical and our precision ceramic machining services.